User:Mr. Ibrahem/Tolbutamide
 | |
| Clinical data | |
|---|---|
| Trade names | Orinase, others |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a682481 |
| License data |
|
| Pregnancy category |
|
| Routes of administration | By mouth (tablet) |
| Drug class | Sulfonylurea[1] |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Protein binding | 96% |
| Metabolism | Liver (CYP2C19-mediated) |
| Elimination half-life | 4.5 to 6.5 hours |
| Excretion | Kidney |
| Identifiers | |
| |
| Chemical and physical data | |
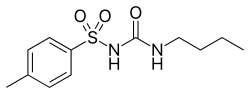
| Formula | C12H18N2O3S |
| Molar mass | 270.35 g·mol−1 |
| 3D model (JSmol) | |
| Melting point | 128.5 to 129.5 °C (263.3 to 265.1 °F) |
| |
| |
| (verify) | |
Tolbutamide is a medication used to treat type 2 diabetes.[1] It is a second line treatment after metformin.[1] It is taken by mouth.[1] Effects may last for up to 24 hrs.[1]
Common side effects include nausea, itchiness, and rash.[1] Other side effect may include low blood sugar.[1] Those with liver or kidney problems have more frequent side effects.[1] Use in pregnancy is not generally recommended.[2] It is a sulfonylurea.[1] It works by stimulating the release of insulin by the pancreas.[1]
Tolbutamide was discovered in 1956 and approved for medical use in 1957.[3][1] It is available as a generic medication.[2] In the United States 100 tablets of 500 mg costs about 92 USD as of 2021.[4] This amount in the United Kingdom costs the NHS about £73.[2]
References[edit]
- ^ a b c d e f g h i j k l m "Tolbutamide Monograph for Professionals". Drugs.com. Archived from the original on 17 August 2019. Retrieved 5 October 2021.
- ^ a b c BNF (80 ed.). BMJ Group and the Pharmaceutical Press. September 2020 – March 2021. p. 749. ISBN 978-0-85711-369-6.
{{cite book}}: CS1 maint: date format (link) - ^ Walker SR (2012). Trends and Changes in Drug Research and Development. Springer Science & Business Media. p. 109. ISBN 9789400926592. Archived from the original on 2017-09-10. Retrieved 2020-11-29.
- ^ "Tolbutamide Prices, Coupons & Patient Assistance Programs". Drugs.com. Archived from the original on 17 April 2021. Retrieved 5 October 2021.
