User:Mr. Ibrahem/Oxaprozin
 | |
| Clinical data | |
|---|---|
| Trade names | Daypro, Dayrun, Duraprox, others |
| Other names | Oxaprozinum[1] |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a693002 |
| Pregnancy category |
|
| Routes of administration | By mouth |
| Drug class | Nonsteroidal anti-inflammatory drug (NSAID)[2] |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | 95% |
| Protein binding | 99% |
| Metabolism | Liver—65% oxidation and 35% glucuronic acid conjugation. 5% are active phenolic metabolites. |
| Elimination half-life | 54.9 hours |
| Identifiers | |
| |
| Chemical and physical data | |
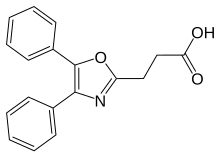
| Formula | C18H15NO3 |
| Molar mass | 293.322 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
Oxaprozin, sold under the brand name Daypro among others, is a nonsteroidal anti-inflammatory drug (NSAID), used to treat inflammation in conditions such as osteoarthritis and rheumatoid arthritis.[2] It is taken by mouth.[2]
Common side effects include abdominal pain, stomach ulcers, gastrointestinal bleeding, nausea, poor kidney function, and itchiness.[2] Other side effects may include liver problems, anaphylaxis, heart failure, myocardial infarction, and Stevens-Johnson syndrome.[2] Use in the later part of pregnancy may harm the baby.[2]
Oxaprozin was patented in 1967 and approved for medical use in 1983.[3] In the United States it costs about 2.50 USD per 600 mg dose as of 2021.[2] It is also available in Canada and Japan.[1]
References[edit]
- ^ a b Society, Swiss Pharmaceutical (2000). Index Nominum 2000: International Drug Directory. Taylor & Francis. p. 768. ISBN 978-3-88763-075-1. Archived from the original on 10 November 2021. Retrieved 10 November 2021.
- ^ a b c d e f g h i "Oxaprozin Prices, Coupons & Patient Assistance Programs". Drugs.com. Archived from the original on 18 January 2021. Retrieved 10 November 2021.
- ^ Fischer, Jnos; Ganellin, C. Robin (2006). Analogue-based Drug Discovery. John Wiley & Sons. p. 520. ISBN 9783527607495. Archived from the original on 2020-08-06. Retrieved 2020-10-17.
