User:Mr. Ibrahem/Modafinil
 | |
| Clinical data | |
|---|---|
| Trade names | Provigil, Alertec, Modavigil, others |
| Other names | CRL-40476; Diphenylmethylsulfinylacetamide |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a602016 |
| License data | |
| Pregnancy category |
|
| Dependence liability | Relatively low |
| Addiction liability | Very low to low[1] |
| Routes of administration | By mouth (tablets)[2] |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | Not determined due to the aqueous insolubility |
| Protein binding | 62.3% |
| Metabolism | Hepatic (primarily via amide hydrolysis;[4] CYP1A2, CYP2B6, CYP2C9, CYP2C19, CYP3A4, CYP3A5 involved [5] |
| Elimination half-life | 15 hours (R-enantiomer), 4 hours (S-enantiomer)[3] |
| Excretion | Urine (80%) |
| Identifiers | |
| |
| Chemical and physical data | |
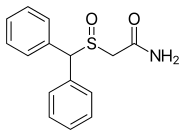
| Formula | C15H15NO2S |
| Molar mass | 273.35 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| (verify) | |
Modafinil, sold under the brand name Provigil among others, is a medication to treat sleepiness due to narcolepsy, shift work sleep disorder, or obstructive sleep apnea.[2][6] While it has seen off-label use as a purported cognitive enhancer, the research on its effectiveness for this use is not conclusive.[8][9] It is taken by mouth.[2]
Common side effects include headache, anxiety, trouble sleeping, and nausea.[2] Serious side effects may include allergic reactions such as anaphylaxis, Stevens–Johnson syndrome, misuse, and hallucinations.[2] It is unclear if use during pregnancy is safe.[2] The amount of medication used may need to be adjusted in those with kidney or liver problems.[2] It is not recommended in those with an arrhythmia, significant hypertension, or left ventricular hypertrophy.[10] How it works is not entirely clear.[2] One possibility is that it may affect the areas of the brain involved with the sleep cycle.[2]
Modafinil was approved for medical use in the United States in 1998.[6] In the United States it is classified as a schedule IV controlled substance.[2] In the United Kingdom it is a prescription only medication.[10] It is available as a generic medication.[10] In the United Kingdom it costs the NHS about £105.21 a month as of 2018.[10] In the United States the wholesale cost per month is about US$34.20 as of 2018.[11] In 2017, it was the 328th most commonly prescribed medication in the United States, with more than 900 thousand prescriptions.[12]
References[edit]
- ^ Mignot EJ (October 2012). "A practical guide to the therapy of narcolepsy and hypersomnia syndromes". Neurotherapeutics. 9 (4): 739–52. doi:10.1007/s13311-012-0150-9. PMC 3480574. PMID 23065655.
Because of the relatively low risk of addiction, modafinil can be more easily prescribed in patients without a clear, biochemically defined central hypersomnia syndrome, and is also easier to stop, if needed. It is also a schedule IV compound.
- ^ a b c d e f g h i j k l m "Modafinil Monograph for Professionals". Drugs.com. American Society of Health-System Pharmacists. Archived from the original on March 30, 2019. Retrieved June 24, 2018.
- ^ "Nuvigil Prescribing Information" (PDF). Nuvigil.com. Archived from the original (PDF) on January 7, 2018. Retrieved January 20, 2019.
- ^ Robertson P, Hellriegel ET (2003). "Clinical pharmacokinetic profile of modafinil". Clinical Pharmacokinetics. 42 (2): 123–37. doi:10.2165/00003088-200342020-00002. PMID 12537513. S2CID 1266677.
- ^ Robertson P, DeCory HH, Madan A, Parkinson A (June 2000). "In vitro inhibition and induction of human hepatic cytochrome P450 enzymes by modafinil". Drug Metabolism and Disposition. 28 (6): 664–71. PMID 10820139.
- ^ a b c "Provigil Prescribing Information" (PDF). United States Food and Drug Administration. Teva Pharmaceuticals USA, Inc. January 2015. Archived (PDF) from the original on February 17, 2017. Retrieved July 18, 2015.
- ^ "WHOCC - ATC/DDD Index". www.whocc.no. Archived from the original on September 20, 2020. Retrieved September 9, 2020.
- ^ Battleday RM, Brem AK (November 2015). "Modafinil for cognitive neuroenhancement in healthy non-sleep-deprived subjects: A systematic review". European Neuropsychopharmacology. 25 (11): 1865–81. doi:10.1016/j.euroneuro.2015.07.028. PMID 26381811. S2CID 23319688. Archived from the original on August 28, 2021. Retrieved August 5, 2020.
- ^ Meulen, Ruud ter; Hall, Wayne; Mohammed, Ahmed (2017). Rethinking Cognitive Enhancement. Oxford University Press. p. 116. ISBN 9780198727392. Archived from the original on July 14, 2020. Retrieved August 5, 2020.
- ^ a b c d BNF 74 (74 ed.). Pharmaceutical Press. September 2017. p. 468. ISBN 978-0857112989.
- ^ "NADAC as of 2018-06-13". Centers for Medicare and Medicaid Services. Archived from the original on August 7, 2020. Retrieved June 24, 2018.
- ^ "Modafinil - Drug Usage Statistics". ClinCalc. Archived from the original on June 19, 2020. Retrieved April 11, 2020.
