User:Mr. Ibrahem/Mitoxantrone
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Novantrone, Onkotrone, others |
| Other names | Mitozantrone |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a608019 |
| Routes of administration | Mainly intravenous |
| Drug class | Antineoplastic (anthracenedione)[1] |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | n/a |
| Protein binding | 78% |
| Metabolism | Liver (CYP2E1) |
| Elimination half-life | 75 hours |
| Excretion | Kidney |
| Identifiers | |
| |
| Chemical and physical data | |
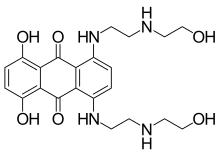
| Formula | C22H28N4O6 |
| Molar mass | 444.488 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| (verify) | |
Mitoxantrone, also known as mitozantrone, is a medication used to treat cancer and multiple sclerosis.[1][2] Types of cancer it is used for include acute myeloid leukemia, prostate cancer, breast cancer, and non-Hodgkin's lymphoma.[1][2] It is given by gradual injection into a vein.[1]
Common side effects include nausea, diarrhea, hair loss, infection, fever, swelling, and shortness of breath.[1] Other side effects may include tumor lysis syndrome, neuropathy, bone marrow suppression, and heart damage.[1] Use in pregnancy may harm the baby.[1] It is an anthracenedione antineoplastic agent.[1]
Mitoxantrone was approved for medical use in the United States in 1987.[1] It is available as a generic medication.[2] In the United Kingdom it costs the NHS about £51 per 20 mg vial as of 2021.[2] This amount in the United States costs about 180 USD.[3]
References[edit]
- ^ a b c d e f g h i j k "MitoXANTRONE Monograph for Professionals". Drugs.com. Archived from the original on 7 May 2021. Retrieved 18 November 2021.
- ^ a b c d BNF 81: March-September 2021. BMJ Group and the Pharmaceutical Press. 2021. p. 948. ISBN 978-0857114105.
- ^ "Mitoxantrone Prices, Coupons & Patient Assistance Programs". Drugs.com. Archived from the original on 19 April 2021. Retrieved 18 November 2021.
