User:Mr. Ibrahem/Lifitegrast
 | |
| Clinical data | |
|---|---|
| Pronunciation | Xiidra /ˈzaɪdrə/[1] |
| Trade names | Xiidra |
| Other names | SAR-1118 |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a616039 |
| Pregnancy category |
|
| Routes of administration | Eye drops |
| Legal status | |
| Legal status | |
| Identifiers | |
| |
| Chemical and physical data | |
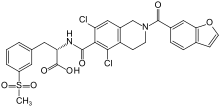
| Formula | C29H24Cl2N2O7S |
| Molar mass | 615.48 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Lifitegrast, sold under the brand name Xiidra, is a medication used to treat dry eyes.[2] It is unclear if its benefits are greater than its harms.[3][4] It is used as eye drops.[2]
Common side effects include blurry vision, red eyes, headache, change in taste, and itchiness.[2] Safety in pregnancy is not clear.[5] It reduces inflammation by inhibiting T cells.[3]
Lifitegrast was approved for medical use in the United States in 2016.[2] It is not approved in either the United Kingdom or Europe.[3][4] In the United States it costs about 580 USD per month as of 2021.[6]
References[edit]
- ^ "Patient information: Xiidra® (ZYE-druh) (lifitegrast ophthalmic solution) 5% for topical ophthalmic use" (PDF). Novartis. June 2020. Archived (PDF) from the original on 2021-02-13. Retrieved 2021-02-05.
- ^ a b c d e f "Lifitegrast Monograph for Professionals". Drugs.com. Archived from the original on 5 March 2021. Retrieved 22 November 2021.
- ^ a b c "Lifitegrast". SPS - Specialist Pharmacy Service. 8 February 2016. Archived from the original on 11 December 2021. Retrieved 23 November 2021.
- ^ a b "Xiidra: Withdrawal of the marketing authorisation application". Archived from the original on 14 November 2021. Retrieved 23 November 2021.
- ^ "Lifitegrast ophthalmic (Xiidra) Use During Pregnancy". Drugs.com. Archived from the original on 28 November 2020. Retrieved 23 November 2021.
- ^ "Lifitegrast Prices, Coupons & Savings Tips - GoodRx". GoodRx. Archived from the original on 28 October 2016. Retrieved 23 November 2021.
