User:Matrix8086/sandbox
 | This is a user sandbox of Matrix8086. You can use it for testing or practicing edits. This is not the sandbox where you should draft your assigned article for a dashboard.wikiedu.org course. To find the right sandbox for your assignment, visit your Dashboard course page and follow the Sandbox Draft link for your assigned article in the My Articles section. |
A chiral auxiliary is a stereogenic group or unit that is temporarily incorporated into an organic compound in order to control the stereochemical outcome of the synthesis.[1][2] The chirality present in the auxiliary can bias the stereoselectivity of one or more subsequent reactions. The auxiliary can then be typically and can be recovered for future use.

Most biological molecules and pharmaceutical targets exist as one of two possible enantiomers; consequently, chemical syntheses of natural products and pharmaceutical agents are frequently designed to obtain the target in enantiomerically pure form.[3] Chiral auxiliaries are one of many strategies available to synthetic chemists to selectively produce the desired stereoisomer of a given compound.[4]
1,1’-Binaphthyl-2,2’-diol (BINOL)[edit]
1,1’-Binaphthyl-2,2’-diol, or BINOL, has been used as chiral auxiliary for the asymmetric synthesis since 1983.[5][6]

Hisashi Yamamoto first utilized (R)-BINOL as a chiral auxiliary in the asymmetric synthesis of limonene, which is a example of cyclic mono-terpenes. (R)-BINOL monoeryl ether was prepared by the monosilylation and alkylation of (R)-BINOL as the chiral auxiliary. Followed with the reduction by organoaluminum reagent, limonene was synthesized with low yields (29% yield) and moderate enantiomeric excesses up to 64% ee. [6]

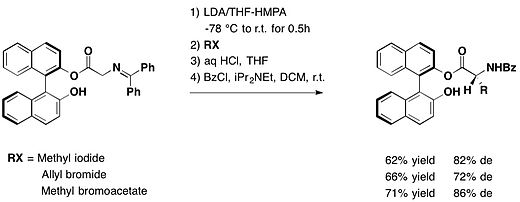
The preparation of a variety of enantiomerically pure uncommon R-amino acids can be achieved by the alkylation of chiral glycine derivatives possessing axially chiral BINOL as an auxiliary. It has been depicted by Fuji et al. Based on different electrophile, the diastereomeric excess varied from 69% to 86.[7]
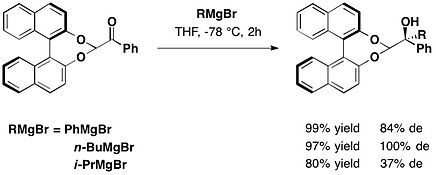
Protected at the aldehyde function with (R)-BINOL, arylglyoxals reacted diastereoselectively with Grignard reagents to afford protected atrolactaldehyde with moderate to excellent diastereomeric excess and high yields.[8]
Trans-2-Phenylcyclohexanol[edit]

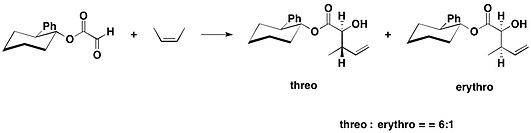
Trans-2-phenylcyclohexanol is one type of chiral auxiliary, which was first introduced by James K. Whitesell and his coworkers in 1985. This chiral auxiliary was used in ene reactions of the derived glyoxylate ester.[9]
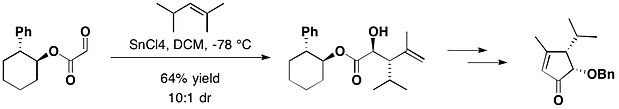
In the total synthesis of (-)-Heptemerone B and (-)-Guanacastepene E, Attached with trans-2-pheynlcyclohexanol, the glyoxylate reacted with 2,4-dimethyl-2-pentene, in the presence of tin(IV) chloride, yielding the desired anti adduct as the major product, together with a small amount of its syn isomer with 10:1 diastereomeric ratio.[10]
Trans-2-cumylcyclohexanol (TCC) has a similar structure to Trans-2-phenylcyclohexanol. In 2015, Brown group published an efficient method in permanganate-mediated oxidative cyclization with this type of chiral auxiliary.[11]

Camphorsultam[edit]
Camphorsultam, or Oppolzer’s sultam, is a typical chiral auxiliary in the asymmetric synthesis.
In the total synthesis of manzacidin B, Ohfune group utilized camphorsultam to construct the core oxazoline ring asymmetrically. Comparing with oxazolidinone as the chiral auxiliary, camphorsultam had a significant (2S,3R)-selectivity. [12]

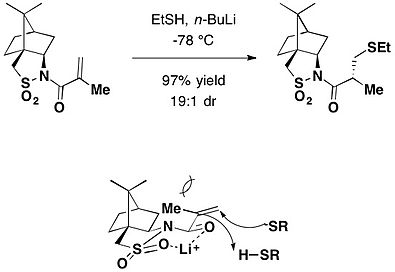
Camphorsultam also acts as a chiral auxiliary in Michael addition. Lithium base promoted stereoselective Michael addition of thiols to N-mcthacryloylcamphorsultam produced the corresponding addition products in high diastereoselectivity.[13]
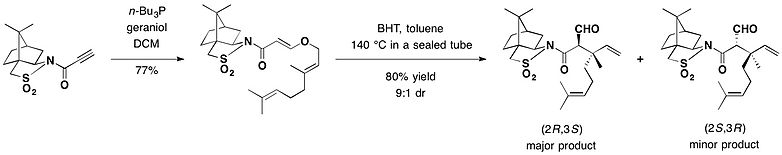
Camphorsultam was used as a chiral auxiliary for the asymmetric Claisen rearrangement. In the presence of butylated hydroxytoluene (BHT) used as a polymerization inhibitor, a toluene solution of the adduct between geraniol and camphorsultam was heated in a sealed tube at 140 °C, to provide mainly the (2R,3S)-isomer as the major rearrangement product in 72% yield, securing the two contiguous stereocenters including the quaternary carbon.[14]
Reference[edit]
- ^ Key Chiral Auxiliary Applications (Second Edition)(ed.: Roos, G.), Academic Press, Boston, 2014. ISBN 978-0-12-417034-6.
- ^ Glorius, F.; Gnas, Y. (2006). "Chiral Auxiliaries — Principles and Recent Applications". Synthesis. 12: 1899–1930. doi:10.1055/s-2006-942399.
- ^ Jamali, Fakhreddin (1993). "Chapter 14: Stereochemically Pure Drugs: An Overview". In Wainer, Irving W. (ed.). Drug Stereochemistry: Analytical Methods and Pharmacology. Marcel Dekker, Inc. pp. 375–382. ISBN 0-8247-8819-2.
- ^ Evans, D. A.; Helmchen, G.; Rüping, M. (2007). "Chiral Auxiliaries in Asymmetric Synthesis". In Christmann, M (ed.). Asymmetric Synthesis — The Essentials. Wiley-VCH Verlag GmbH & Co. pp. 3–9. ISBN 978-3-527-31399-0.
- ^ Brunel, Jean Michel. "BINOL: A Versatile Chiral Reagent". Chemical Reviews. 105 (3): 857–898. doi:10.1021/cr040079g.
{{cite journal}}: no-break space character in|title=at position 7 (help) - ^ a b Sakane, Soichi; Fujiwara, Junya; Maruoka, Keiji; Yamamoto, Hisashi. "Chiral leaving group. Biogenetic-type asymmetric synthesis of limonene and bisabolenes". Journal of the American Chemical Society. 105 (19): 6154–6155. doi:10.1021/ja00357a033.
- ^ Tanaka, Kiyoshi; Ahn, Mija; Watanabe, Yukari; Fuji, Kaoru (1996-06-01). "Asymmetric synthesis of uncommon α-amino acids by diastereoselective alkylations of a chiral glycine equivalent". Tetrahedron: Asymmetry. 7 (6): 1771–1782. doi:10.1016/0957-4166(96)00212-1.
- ^ Maglioli, Paola; De Lucchi, Ottorino; Delogu, Giovanna; Valle, Giovanni (1992-01-01). "Highly diastereoselective reduction and addition of nucleophiles to binaphthol-protected arylglyoxals". Tetrahedron: Asymmetry. 3 (3): 365–366. doi:10.1016/S0957-4166(00)80276-1.
- ^ Buchi, George; Vogel, Dennis E. "A new method for the preparation of .gamma.,.delta.-unsaturated ketones via Claisen rearrangement". The Journal of Organic Chemistry. 50 (23): 4664–4665. doi:10.1021/jo00223a056.
- ^ Miller, Aubry K.; Hughes, Chambers C.; Kennedy-Smith, Joshua J.; Gradl, Stefan N.; Trauner, Dirk. "Total Synthesis of (−)-Heptemerone B and (−)-Guanacastepene E". Journal of the American Chemical Society. 128 (51): 17057–17062. doi:10.1021/ja0660507.
- ^ Al Hazmi, Ali M.; Sheikh, Nadeem S.; Bataille, Carole J. R.; Al-Hadedi, Azzam A. M.; Watkin, Sam V.; Luker, Tim J.; Camp, Nicholas P.; Brown, Richard C. D. "trans -2-Tritylcyclohexanol as a Chiral Auxiliary in Permanganate-Mediated Oxidative Cyclization of 2-Methylenehept-5-enoates: Application to the Synthesis of trans -(+)-Linalool Oxide". Organic Letters. 16 (19): 5104–5107. doi:10.1021/ol502454r.
- ^ Shinada, Tetsuro; Oe, Kentaro; Ohfune, Yasufumi (2012-06-27). "Efficient total synthesis of manzacidin B". Tetrahedron Letters. 53 (26): 3250–3253. doi:10.1016/j.tetlet.2012.04.042.
- ^ Tsai, Wen-Jiuan; Lin, Yi-Tsong; Uang, Biing-Jiun (1994-07-01). "Asymmetric Michael addition of thiols to (1R,2R,4R)-(−)-2,10-N-enoylcamphorsultam". Tetrahedron: Asymmetry. 5 (7): 1195–1198. doi:10.1016/0957-4166(94)80155-X.
- ^ Takao, Ken-ichi; Sakamoto, Shu; Touati, Marianne Ayaka; Kusakawa, Yusuke; Tadano, Kin-ichi (2012-11-08). "Asymmetric Construction of All-Carbon Quaternary Stereocenters by Chiral-Auxiliary-Mediated Claisen Rearrangement and Total Synthesis of (+)-Bakuchiol". Molecules. 17 (11): 13330–13344. doi:10.3390/molecules171113330.
{{cite journal}}: CS1 maint: unflagged free DOI (link)
