From Wikipedia, the free encyclopedia
This is
the user sandbox of Darktin . A user sandbox is a subpage of the user's
user page . It serves as a testing spot and page development space for the user and is
not an encyclopedia article .
Create or edit your own sandbox here . Other sandboxes: Main sandbox | Template sandbox
Submit your draft for review!
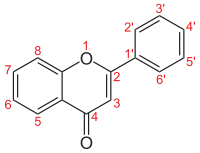
Flavones and their structure [1]
Name
Structure
R3
R5
R6
R7
R8
R2'
R3'
R4'
R5'
R6'
Flavone
–
–
–
–
–
–
–
–
–
–
Primuletin
–
–OH
–
–
–
–
–
–
–
–
Chrysin
–
–OH
–
–OH
–
–
–
–
–
–
Tectochrysin
–
–OH
–
–OCH3
–
–
–
–
–
–
Primetin
–
–OH
–
–
–OH
–
–
–
–
–
Apigenin
–
–OH
–
–OH
–
–
–
–OH
–
–
Acacetin
–
–OH
–
–OH
–
–
–
–OCH3
–
–
Genkwanin
–
–OH
–
–OCH3
–
–
–
–OH
–
–
Echioidinin
–
–OH
–
–OCH3
–
–OH
–
–
–
–
Baicalein
–
–OH
–OH
–OH
–
–
–
–
–
–
Oroxylon
–
–OH
–OCH3
–OH
–
–
–
–
–
–
Negletein
–
–OH
–OH
–OCH3
–
–
–
–
–
–
Norwogonin
–
–OH
–
–OH
–OH
–
–
–
–
–
Wogonin
–
–OH
–
–OH
–OCH3
–
–
–
–
–
Geraldone
–
–
–
–OH
–
–
–OCH3
–OH
–
–
Tithonine
–
–
–
–OCH3
–
–
–OH
–OCH3
–
–
Luteolin
–
–OH
–
–OH
–
–
–OH
–OH
–
–
Chrysoeriol
–
–OH
–
–OH
–
–
–OCH3
–OH
–
–
Diosmetin
–
–OH
–
–OH
–
–
–OH
–OCH3
–
–
Pilloin
–
–OH
–
–OCH3
–
–
–OH
–OCH3
–
–
Velutin
–
–OH
–
–OCH3
–
–
–OCH3
–OH
–
–
Norartocarpetin
–
–OH
–
–OH
–
–OH
–
–OH
–
–
Artocarpetin
–
–OH
–
–OCH3
–
–OH
–
–OH
–
–
Scutellarein
–
–OH
–OH
–OH
–
–
–
–OH
–
–
Hispidulin
–
–OH
–OCH3
–OH
–
–
–
–OH
–
–
Sorbifolin
–
–OH
–OH
–OCH3
–
–
–
–OH
–
–
Pectolinarigenin
–
–OH
–OCH3
–OH
–
–
–
–OCH3
–
–
Cirsimaritin
–
–OH
–OCH3
–OCH3
–
–
–
–OH
–
–
Mikanin
–
–OH
–OCH3
–OCH3
–
–
–
–OCH3
–
–
Isoscutellarein
–
–OH
–
–OH
–OH
–
–
–OH
–
–
Zapotinin
–
–OH
–OCH3
–
–
–OCH3
–
–
–
–OCH3
Zapotin
–
–OCH3
–OCH3
–
–
–OCH3
–
–
–
–OCH3
Cerrosillin
–
–OCH3
–OCH3
–
–
–
–OCH3
–
–OCH3
–
Alnetin
–
–OH
–OCH3
–OCH3
–OCH3
–
–
–
–
–
Tricetin
–
–OH
–
–OH
–
–
–OH
–OH
–OH
–
Tricin
–
–OH
–
–OH
–
–
–OCH3
–OH
–OCH3
–
Corymbosin
–
–OH
–
–OCH3
–
–
–OCH3
–OCH3
–OCH3
–
Nepetin
–
–OH
–OCH3
–OH
–
–
–OH
–OH
–
–
Pedalitin
–
–OH
–OH
–OCH3
–
–
–OH
–OH
–
–
Nodifloretin
–
–OH
–OH
–OH
–
–
–OCH3
–OH
–
–
Jaceosidin
–
–OH
–OCH3
–OH
–
–
–OCH3
–OH
–
–
Cirsiliol
–
–OH
–OCH3
–OCH3
–
–
–OH
–OH
–
–
Eupatilin
–
–OH
–OCH3
–OH
–
–
–OCH3
–OCH3
–
–
Cirsilineol
–
–OH
–OCH3
–OCH3
–
–
–OCH3
–OH
–
–
Eupatorin
–
–OH
–OCH3
–OCH3
–
–
–
–OCH3
–OH
–
Sinensetin
–
–OCH3
–OCH3
–OCH3
–
–
–
–OCH3
–OCH3
–
Hypolaetin
–
–OH
–
–OH
–OH
–
–OH
–OH
–
–
Onopordin
–
–OH
–
–OH
–OCH3
–
–OH
–OH
–
–
Wightin
–
–OH
–
–OCH3
–OCH3
–OCH3
–OH
–
–
–
Nevadensin
–
–OH
–OCH3
–OH
–OCH3
–
–
–OCH3
–
–
Xanthomicrol
–
–OH
–OCH3
–OCH3
–OCH3
–
–
–OH
–
–
Tangeretin
–
–OCH3
–OCH3
–OCH3
–OCH3
–
–
–OCH3
–
–
Serpyllin
–
–OH
–
–OCH3
–OCH3
–OCH3
–OCH3
–OCH3
–
–
Sudachitin
–
–OH
–OCH3
–OH
–OCH3
–
–OCH3
–OH
–
–
Acerosin
–
–OH
–OCH3
–OH
–OCH3
–
–OH
–OCH3
–
–
Hymenoxin
–
–OH
–OCH3
–OH
–OCH3
–
–OCH3
–OCH3
–
–
Gardenin D
–
–OH
–OCH3
–OCH3
–OCH3
–
–OH
–OCH3
–
–
Nobiletin
–
–OCH3
–OCH3
–OCH3
–OCH3
–
–OCH3
–OCH3
–
–
Scaposin
–
–OH
–OCH3
–OH
–OCH3
–
–OCH3
–OCH3
–OH
–
Name
Struktur
R3
R5
R6
R7
R8
R2'
R3'
R4'
R5'
R6'