User:Mr. Ibrahem/Phendimetrazine
 | |
| Clinical data | |
|---|---|
| Trade names | Bontril, Adipost, Anorex-SR, others |
| AHFS/Drugs.com | Monograph |
| Pregnancy category |
|
| Routes of administration | By mouth |
| Drug class | Amphetamine[1] |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | Peak plasma levels occur within 1 to 3 hours. Absorption is usually complete by 4 to 6 hours |
| Metabolism | Liver |
| Elimination half-life | 19-24 hours |
| Excretion | Urinary elimination |
| Identifiers | |
| |
| Chemical and physical data | |
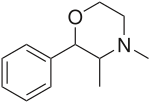
| Formula | C12H17NO |
| Molar mass | 191.274 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| (verify) | |
Phendimetrazine, sold under the brand name Bontril among others, is a medication used to treat obesity.[1] Use is only recommended for a few weeks.[1] It is used together with dieting and exercise.[1][2] It is taken by mouth.[1]
Common side effects include palpitations, fast heart rate, high blood pressure, agitation, trouble sleeping, headache, psychosis, and diarrhea.[1] Other side effects may include abuse, valvular heart disease, and pulmonary hypertension.[1] Use during pregnancy may harm the baby.[3] It is an amphetamine which works by decreasing appetite.[1]
Phendimetrazine was approved for medical use in the United States in 1975.[1] Europe voted to removal approval in 1999.[4] It is available as a generic medication.[2] In the United States 60 tablets of 35 mg costs about 10 USD as of 2021.[2] In the United States it is a Schedule III controlled substance.[5]
References[edit]
- ^ a b c d e f g h i j k "Phendimetrazine Monograph for Professionals". Drugs.com. Archived from the original on 27 January 2021. Retrieved 27 October 2021.
- ^ a b c "Phendimetrazine Prices, Coupons & Savings Tips - GoodRx". GoodRx. Archived from the original on 6 February 2017. Retrieved 27 October 2021.
- ^ "Phendimetrazine Use During Pregnancy". Drugs.com. Archived from the original on 5 December 2020. Retrieved 27 October 2021.
- ^ "https://www.ema.europa.eu/en/documents/press-release/extraordinary-meeting-finalise-review-anorectic-agents_en.pdf" (PDF). Archived (PDF) from the original on 28 October 2021. Retrieved 27 October 2021.
{{cite web}}: External link in|title= - ^ "PART 1308 - Section 1308.13 Schedule III". www.deadiversion.usdoj.gov. Archived from the original on 18 October 2021. Retrieved 27 October 2021.
