User:Mr. Ibrahem/Ivabradine
 | |
 | |
| Clinical data | |
|---|---|
| Pronunciation | /ɪˈvæbrədiːn/ |
| Trade names | Corlanor, Procoralan |
| Other names | Ivabradine hydrochloride, S-16257 |
| AHFS/Drugs.com | Monograph |
| License data |
|
| Routes of administration | By mouth |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | 40% |
| Protein binding | 70% |
| Metabolism | Liver (first-pass) >50%, CYP3A4-mediated |
| Elimination half-life | 2 hours |
| Excretion | Kidney and fecal |
| Identifiers | |
| |
| Chemical and physical data | |
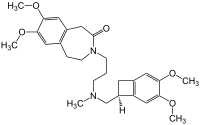
| Formula | C27H36N2O5 |
| Molar mass | 468.594 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
Ivabradine, sold under the brand name Procoralan among others, is a medication used for long term stable heart-related chest pain and heart failure.[1] It may be used when the heart rate is at least 70 beats per minute and the condition is not fully managed by beta blockers.[1][2] It is taken by mouth.[3]
Common side effects include temporarily seeing brightness and slow heart rate.[1] Other side effects may include atrial fibrillation and angioedema.[3] Use during pregnancy may harm the baby.[3] It works by blocking If current in the sinus node which lowers the heart rate.[1]
Ivabradine was approved for medical use in Europe in 2005 and the United States in 2015.[1][3] It is available as a generic medication.[4][5] In the United Kingdom 4 weeks of medication costs the NHS about £5.[4] In the United States this amount costs about 450 USD.[6]
References[edit]
- ^ a b c d e f g h "Procoralan". Archived from the original on 10 November 2021. Retrieved 1 December 2021.
- ^ Yancy, CW; Jessup, M; Bozkurt, B; Butler, J; Casey, DE Jr; Colvin, MM; Drazner, MH; Filippatos, G; Fonarow, GC; Givertz, MM; Hollenberg, SM; Lindenfeld, J; Masoudi, FA; McBride, PE; Peterson, PN; Stevenson, LW; Westlake, C (20 May 2016). "2016 ACC/AHA/HFSA Focused Update on New Pharmacological Therapy for Heart Failure: An Update of the 2013 ACCF/AHA Guideline for the Management of Heart Failure: A Report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines and the Heart Failure Society of America". Circulation. 134 (13): e282–e293. doi:10.1161/CIR.0000000000000435. PMID 27208050.
- ^ a b c d "Ivabradine Monograph for Professionals". Drugs.com. Archived from the original on 21 January 2021. Retrieved 26 November 2021.
- ^ a b BNF 81: March-September 2021. BMJ Group and the Pharmaceutical Press. 2021. p. 227. ISBN 978-0857114105.
- ^ Research, Center for Drug Evaluation and (10 February 2022). "2021 First Generic Drug Approvals". FDA. Archived from the original on 21 June 2022. Retrieved 22 October 2022.
- ^ "Ivabradine Prices, Coupons & Savings Tips - GoodRx". GoodRx. Retrieved 1 December 2021.
