User:Mr. Ibrahem/Hydrocortisone
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | A-hydrocort, Cortef, Solu-cortef, others[1] |
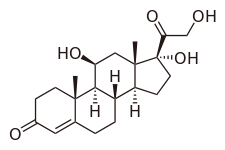
| Other names | Cortisol; 11β,17α,21-Trihydroxypregn-4-ene-3,20-dione |
| AHFS/Drugs.com | Systemic: Monograph Topical: Monograph Eye and ear: Monograph |
| MedlinePlus | a682206 |
| License data |
|
| Pregnancy category |
|
| Routes of administration | By mouth (tablets), intravenous, topical, rectal |
| Drug class | Corticosteroid; glucocorticoid; mineralocorticoid |
| Legal status | |
| Legal status | |
| Identifiers | |
| |
| Chemical and physical data | |
| Formula | C21H30O5 |
| Molar mass | 362.466 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
Hydrocortisone is the name for the hormone cortisol when supplied as a medication.[4] Uses include conditions such as adrenocortical insufficiency, adrenogenital syndrome, high blood calcium, thyroiditis, rheumatoid arthritis, dermatitis, asthma, and COPD.[1] It is the treatment of choice for adrenocortical insufficiency.[5] It can be given by mouth, topically, or by injection.[1] Stopping treatment after long-term use should be done slowly.[1]
Side effects may include mood changes, increased risk of infection, and swelling.[1] With long-term use common side effects include osteoporosis, upset stomach, physical weakness, easy bruising, and yeast infections.[1] While used, it is unclear if it is safe during pregnancy.[6] Hydrocortisone is a glucocorticoid and works as an anti-inflammatory and by immune suppression.[1]
Hydrocortisone was patented in 1936 and approved for medical use in 1941.[7][8] It is on the World Health Organization's List of Essential Medicines.[9] It is available as a generic medication.[1] The wholesale cost in the developing world is about US$0.27 per day as of 2014 for the form taken by mouth.[10] In the United States, it costs less than US$25 for a typical month of treatment.[5] In 2017, it was the 154th most commonly prescribed medication in the United States, with more than four million prescriptions.[11][12]
References[edit]
- ^ a b c d e f g h "Hydrocortisone". Drugs.com. American Society of Health-System Pharmacists. February 9, 2015. Archived from the original on 20 September 2016. Retrieved 30 August 2016.
- ^ "HYDROCORTISONE injectable - Essential drugs". medicalguidelines.msf.org. Archived from the original on 28 August 2021. Retrieved 2 September 2020.
- ^ "WHOCC - ATC/DDD Index". www.whocc.no. Archived from the original on 19 August 2020. Retrieved 2 September 2020.
- ^ Becker, Kenneth L. (2001). Principles and Practice of Endocrinology and Metabolism. Lippincott Williams & Wilkins. p. 762. ISBN 9780781717502. Archived from the original on 2016-09-14.
- ^ a b Hamilton, Richart (2015). Tarascon Pocket Pharmacopoeia 2015 Deluxe Lab-Coat Edition. Jones & Bartlett Learning. p. 202. ISBN 9781284057560.
- ^ "Hydrocortisone Pregnancy and Breastfeeding Warnings". Drugs.com. Archived from the original on 20 September 2016. Retrieved 1 September 2016.
- ^ U.S. patent 2,183,589
- ^ Fischer, Jnos; Ganellin, C. Robin (2006). Analogue-based Drug Discovery. John Wiley & Sons. p. 484. ISBN 9783527607495. Archived from the original on 2019-03-02. Retrieved 2019-03-01.
- ^ World Health Organization (2019). World Health Organization model list of essential medicines: 21st list 2019. Geneva: World Health Organization. hdl:10665/325771. WHO/MVP/EMP/IAU/2019.06. License: CC BY-NC-SA 3.0 IGO.
- ^ "Hydrocortisone". International Drug Price Indicator Guide. Archived from the original on 27 August 2018. Retrieved 1 September 2016.
- ^ "The Top 300 of 2020". ClinCalc. Archived from the original on 12 February 2021. Retrieved 11 April 2020.
- ^ "Hydrocortisone - Drug Usage Statistics". ClinCalc. Archived from the original on 12 April 2020. Retrieved 11 April 2020.
