User:Mr. Ibrahem/Cangrelor
 | |
| Clinical data | |
|---|---|
| Trade names | Kengreal, Kengrexal, Canreal |
| Other names | AR-C69931MX |
| AHFS/Drugs.com | Monograph |
| License data | |
| Routes of administration | Intravenous |
| Drug class | P2Y12 inhibitor[1] |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | 100% (IV) |
| Protein binding | ~97–98%. |
| Metabolism | Rapid deactivation in the circulation (independent of CYP system) |
| Elimination half-life | ~3–6 minutes |
| Excretion | Kidney (58%), Bile duct (35%) |
| Identifiers | |
| |
| Chemical and physical data | |
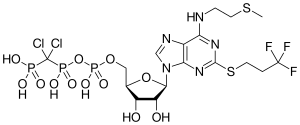
| Formula | C17H25Cl2F3N5O12P3S2 |
| Molar mass | 776.35 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
Cangrelor, sold under the brand name Kengreal among others, is a medication used in those undergoing percutaneous coronary intervention (PCI) to prevent blood clots.[2] It is used with aspirin.[2] It is given by injection into a vein.[1]
Common side effects include bleeding and shortness of breath.[2] Other side effects may include allergic reactions.[2] Safety in pregnancy is unclear.[1] It is a P2Y12 inhibitor which blocks platelets from sticking together.[1][2]
Cangrelor was approved for medical use in the United States and Europe in 2015.[1][2] In the United Kingdom 50 mg costs the NHS about £250 as of 2021.[3] This amount in the United States costs about 830 USD.[4]
References[edit]
- ^ a b c d e "Cangrelor Monograph for Professionals". Drugs.com. Archived from the original on 21 January 2021. Retrieved 29 December 2021.
- ^ a b c d e f g h "Kengrexal". Archived from the original on 17 April 2021. Retrieved 29 December 2021.
- ^ BNF 81: March-September 2021. BMJ Group and the Pharmaceutical Press. 2021. p. 225. ISBN 978-0857114105.
- ^ "Kengreal Prices, Coupons & Patient Assistance Programs". Drugs.com. Archived from the original on 17 April 2021. Retrieved 29 December 2021.
